A fluorinated polymer with a high dielectric constant is demonstrated within thin-film transistors. Crosslinking polymer chains reduces energetic disorder at the insulator-semiconductor interface, resulting in an increase in the charge mobility of rubrene single-crystal field-effect transistors. Image: Enrique Gomez / Penn State
Controlling chain conformations to enhance electronic devices
12/01/2016
UNIVERSITY PARK, Pa. — Controlling the way fluorinated polymer chains twist and turn may enable fast and flexible electrical circuits, according to collaborative research conducted at Penn State. The findings may offer substantial impact on the development of new polymer-type materials used in flexible electronic applications.
Organic thin film transistors (OTFTs), which integrate organic semiconducting compounds in electronic components, have the potential to revolutionize the field of flexible electronics by generating bendable and foldable devices that act as electronic circuits. OTFTs are the building blocks of more complicated circuits and serve as the “on” and “off” switches between various electronic states. They are comprised of multiple layers, including a dielectric layer—which functions as an insulator—responsible for inducing charge.
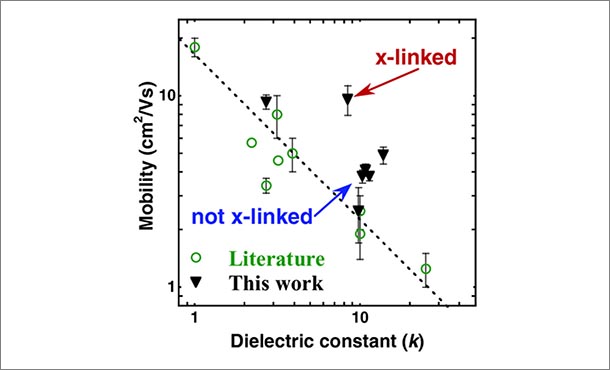
“Our work aims to introduce new, more stable polymers into electrical circuits without negatively impacting performance,” said Enrique Gomez, associate professor of chemical engineering, Penn State. “By controlling the way that polymer chains twist and bend at a molecular level in OTFTs, we have found that we are able to break the universal relationship between charge mobility of the semiconductor and charge storage capacity of the dielectric layer, providing a basis to create fast and reliable circuits from organic materials.”
Controlling the order and arrangement of polymer chains along the semiconductor interface is the key in breaking the relationship, according to research published in Advanced Materials.
Polymers are capable of storing large amounts of electrical energy in the insulator, however, it is known that electrical energy slows as it travels through semiconducting materials. The hope is that new findings about the inverse relationship will lead to the development of fast organic devices with the capacity to revolutionize the flexible electronics industry.
Controlled arrangement of polymers is possible using new chemistries developed at Penn State that introduce cross-linkable polymers into high-charge capacity chains.
Gomez explained, “Polymers can easily be imagined as long macromolecules that resemble spaghetti noodles. They either have the appearance of cooked spaghetti, where all of the chains are tangled and randomly entwined, or uncooked spaghetti, in which the chains are straight and neatly stacked, providing order along the backbone.”
Crosslinking polymer chains creates an alignment of polymers along the insulating level of OTFTs. The alignment leads to a decrease in charge traps—barriers or impediments along the insulator”ultimately, reducing current leakage and producing faster and more efficient circuits.
“We can think of charge traps as pot holes in the highway that electrons must travel over,” said Gomez. “Decreasing the frequency of charge traps increases the speed in which OTFTs can switch on and off.”
For this project, researchers chose to focus on fluorinated polymers, due to their exceptional stability and high propensity to store energy, and transistors derived from rubrene, an aromatic hydrocarbon.
Final results demonstrated that cross-linked insulators increased the mobility in OTFTs by more than one order of magnitude—about 10 times—higher than that of rubrene single-crystal devices made with conventional insulators.
The researchers are hopeful that the results will lead to the discovery of new low-cost materials that can be used in flexible electronic devices, which encompass a myriad of applications, including digital displays, wearable technologies and health monitoring equipment, among many others.
Collaborating with Gomez on the work were fellow Penn State researchers Jwala Adhikari, primary author and recent Penn State chemical engineering Ph.D.; Qing Wang, professor of materials science and engineering; Matthew Gadinski, graduate student in the Department of Materials Science and Engineering; Qi Li, postdoctoral fellow in the Department of Materials Science and Engineering; Thomas Jackson, Robert E. Kirby Chair Professor of Electrical Engineering; and Kaige G. Sun, a recent Penn State electrical engineering Ph.D.
Additional collaborators included Alejandro L. Briseno, professor of polymer science and engineering, University of Massachusettes, Amherst; Marcos Reyes-Martinez, postdoctoral research associate in Chemical and Biological Engineering, Princeton University; and Elissei Iagodkine, associate research scientist, Dow Chemical Company.
The Dow Chemical Company funded this work through a grant established to advance flexible and printed electronics at Penn State.



